Electrolysis
|
Electrolysis is when a chemical bond in a compound breaks by passing an electric current through them
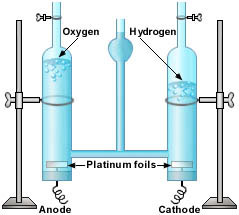
Electrolysis of water involved the breaking down of a water molecule. Since 2H2O --> 2H2 + O2 ( 2 water molecules equal 4 hydrogen atoms and 2 oxygen atoms) we can identify that twice as much hydrogen gas is produced than oxygen gas. Hydrogen gas is H2 while Oxygen gas is O2. Therefore to balance the equation : H2O --> H2 + O2, we must double the amount of water molecules which will give us: 2H2O --> 2H2 +O2. An electric power source is connected to 2 electrodes. These two electrodes are Anode (positive) and Cathode (negative). Hydrogen gas always appears at the negative electrode while oxygen always appears at the positive electrode. The diagram on the right is electrolysis of water. You can also identify and differentiate between hydrogen and oxygen gas by looking at each tube carefully. You will notice that the tube with the most gas in it is labelled hydrogen. This proves the theory that twice as much hydrogen gas is produced that oxygen gas. |