Thermal Decomposition
|
Thermal decomposition is when one reactant produces two products and one of these is usually a gas and other is usually a metal oxide.
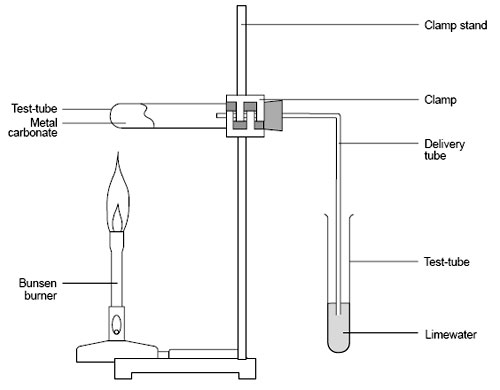
e.g. zinc carbonate --> zinc oxide carbon dioxide On the diagram on the right, you can see that a metal carbonate is being heated using a Bunsen burner. Also you can see that the test tube is connected to another test tube that has lime water in it. This is to test which gas has been released. If carbon dioxide is released, then the lime water will turn cloudy. |